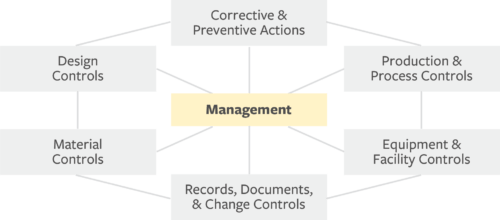
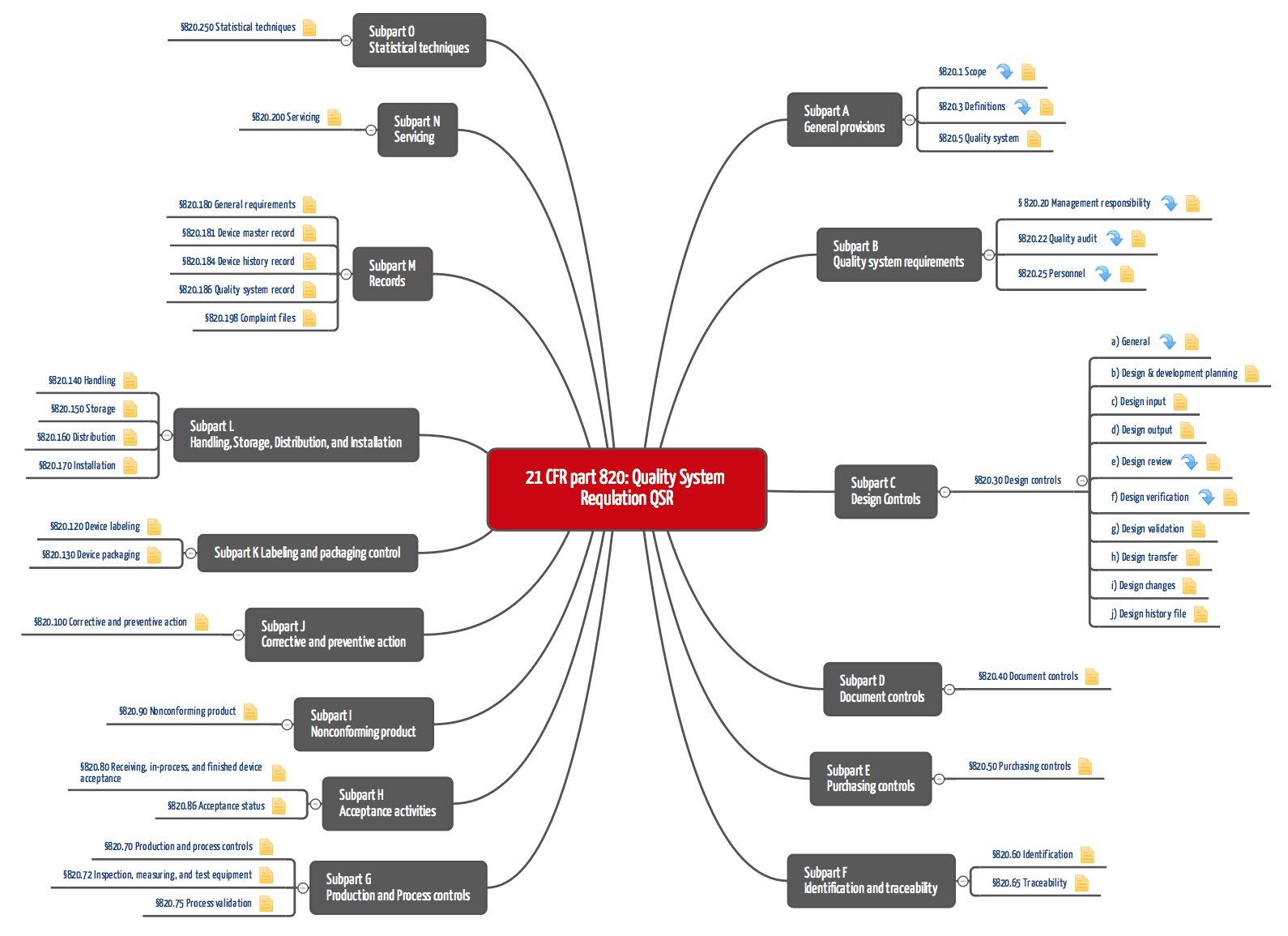
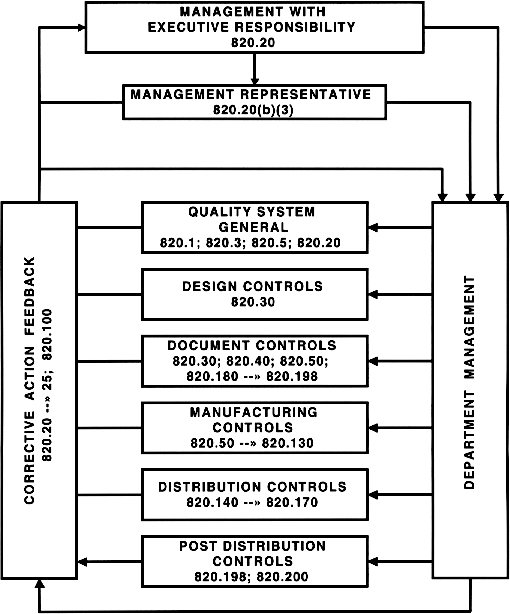
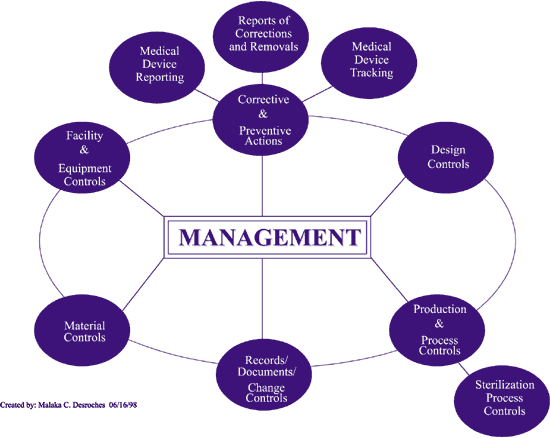
Operon Strategist - FDA 21 CFR Part 820 Quality System Regulation Consultant Services for Medical Device Manufacturers. US FDA QSR, GMP Guidelines, FDA (510k), CE Mark Regulatory. https://operonstrategist.com/services/turnkey-project/quality-management ...